Liu Yu's group’s research result about “Controlled Release And Bound of Guest Molecules by Synthesized Photo- And Thermal-Responsive Host Macrocycles” pubilished on JACS
Allostery is the condition of a protein in which the structure and activity are triggered by external stimuli, to perform the corresponding function. In order to mimic nature’s complex allosteric processes, the design and synthesis of well-crafted artificial macrocycles have long been a worthwhile goal in supramolecular chemistry. However, photo-isomerized small molecules may reduce or lose their configurational interconversion properties under actions of ring tension or internal stress. Therefore, constructing light-responsive rigid macrocyclic host molecules has always been a tough problem in the field of supramolecular.
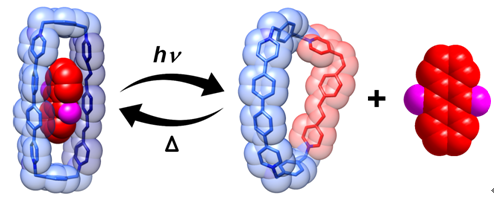
Recently, Yu Liu group (State Key Laboratory of Elemento-Organic Chemistry, Nankai University), developed a host semi-rigid photo- and thermal-responsive tetracationic organic cyclophane (OPVEx2Box4+), which has been shown to be interconverted reversibly between (EE)- and (EZ)-configurations by alternating light irradiation and heating. Due to the effect of macrocyclic tension, the reversibility of isomeric reaction of the cyclophane is further improved. Benefitted from the allosteric regulation of OPVEx2Box4+, a wide array of electron-rich and electron-deficient aromatic guest molecules can be released and bound reversibly. The photo- and thermal-controlled molecular encapsulation strategy not only provides the incentive to design more-advanced photo-responsive host-guest systems and switchable molecular machines but also possesses the potential for applications in the fields of active ingredients stabilization, controllable drug release, and toxin separations.
Relating work has been published on Journal of American Chemical Society (JACS), and selected as the cover paper.
(See: J. Am. Chem. Soc., 2019, 141, 1280,https://pubs.acs.org.ccindex.cn/doi/10.1021/jacs.8b10526)

