Work of Zi Weiwei Research Group Published in JACS: Synergistic Pd/Amine-Catalyzed Stereodivergent
Zi Weiwei research group achieved the first stereodivergent hydroalkylation reactions of 1,3-dienes with aldehydes using a synergistic palladium/amine catalyst system. The study is published in J. Am. Chem. Soc. (https://doi.org/10.1021/jacs.1c02220).
Metal-hydride-catalyzed hydroalkylation of 1,3-dienes with enolizable carbonyl compounds is a powerful method for asymmetric construction of C–C bonds with high atom and step economy. PdH-catalyzed and NiH-catalyzed hydroalkylation reactions of 1,3-dienes with 1,3-dicarbonyl compounds, ketones and amides were already achieved. Although their enantioselectivity can be well controlled, their diastereoselectivity is yet to be satisfactory when two stereocenters are involved. Meanwhile, aldehydes have not yet been used for the direct hydroalkylation of 1,3-dienes because they are less stable than other carbonyl compounds under basic conditions and they have the potential to rapidly epimerize at the α-position.
Zi Weiwei research group has been devoted to the coupling reaction catalyzed by PdH for a long time. Recently, they have realized the first method for stereodivergent hydroalkylation reactions between 1,3-dienes and aldehydes, with synergistic catalysis by a chiral palladium compound and a chiral amine. This method provides highly diastereo- and enantioselective access to all four diastereomers of chiral aldehyde products with α,β-vicinal stereocenters. Isotope experiment and density functional theory calculations confirmed that the mechanism involved PdH formation and subsequent migratory insertion into the alkene, followed by C–C bond formation. The two newly formed stereocenters were independently controlled by the Pd and amine catalysts, and distortion/interaction analysis revealed that distortion energies rather than interaction energies are the primary determinants of the stereoselectivity.
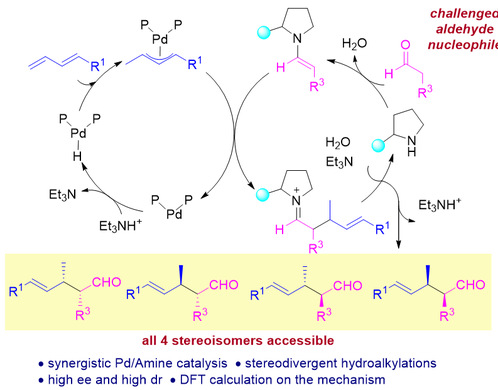
Figure 1. Stereodivergent Hydroalkylation Reactions with Catalysis by Pdh/Amine
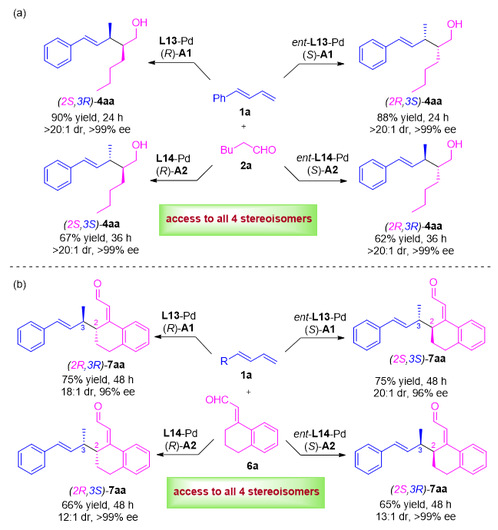
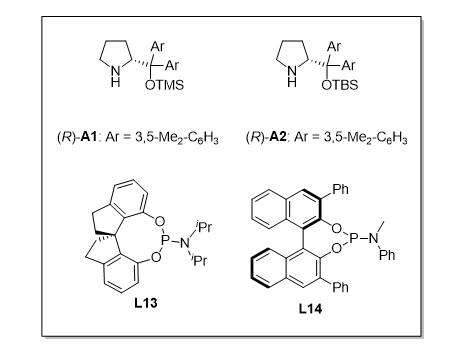
Figure 2. Demonstration of Stereodivergent Synthesis
The work was recently published in full in JACS. Doctoral student Wang Hongfa is the main author of this paper and the first author of this paper. Zhang Ruiyuan, the second author of this paper, has provided some assistance with respect to substrate development. Dr. Zhang Qinglong assisted in guiding the work. The research was supported by the National Natural Science Foundation of China, Foundation of Postdoctoral and Nankai University.

