Nankai University Team Makes Key Breakthrough in Ultra-High Specific Energy Batteries Published in Nature
Tianjin, February 26th, 2023 - The latest research progress in high-energy-density lithium batteries by Professor Qing Zhao from the College of Chemistry at Nankai University, Academician Jun Chen, Vice President of Nankai University and Director of the National Key Laboratory of Special Chemical Power Sources, and Researcher Yong Li from Shanghai Institute of Space Power Sources has been published online in the prestigious academic journal Nature. (Reported by ********, Photos by ********)

The research is titled Hydrofluorocarbon electrolytes for energy-dense and low-temperature batteries. The team conducted research on lithium battery electrolytes, breaking the long-standing kinetic constraints of oxygen coordination in electrolytes. They designed and synthesized a series of new fluorinated hydrocarbon solvent molecules with fluorine coordination. Based on this, they constructed an electrolyte system that replaced the traditional lithium-oxygen coordination in electrolytes, enabling a 700 Wh/kg lithium metal battery to achieve reversible cycling.
Since the invention of the voltaic pile (Voltaic battery) by Volta in 1800, batteries have permeated every aspect of our lives. From lead-acid batteries, nickel-metal-hydride batteries to lithium-ion batteries, which are called the energy heart of modern society, the evolution of battery systems is inseparable from the renewal and iteration of electrolytes. For a long time, oxygen atoms have been regarded as indispensable elements in the solvents of electrolytes. For instance, the current commercial lithium-ion battery electrolytes are usually composed of lithium salts and carbonate solvents. The ion-dipole interaction between lithium and oxygen in the carbonate solvents can promote the dissolution of lithium salts. However, this solvent has poor wettability and requires a large amount, which makes it difficult to further increase the energy density of the battery; at the same time, the strong interaction hinders the interfacial charge transfer in the battery and limits the low-temperature performance, usually the battery cannot work below-50℃.

Challenging Traditional Electrolyte Chemistry
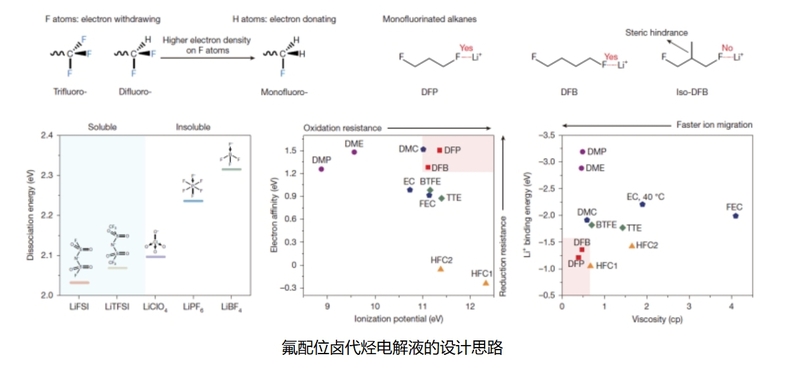
Conventional electrolytes for electrochemical devices primarily rely on oxygen (O)- and nitrogen (N)-based solvents. While these systems facilitate ion dissociation and transport through dipole-ion interactions with metal cations (e.g., Li+, Na+), they hinder charge transfer at the electrolyte-electrode interface. The Nankai team’s fluorine-based ligands, designed with precise steric hindrance and Lewis basicity, dissolve salts at concentrations exceeding 2 mol/L—a marked improvement over traditional solvents.

Key Advancements
1,3-Difluoropropane (DFP) Electrolyte: This novel solvent achieves ultra-low viscosity (0.95 cp), high oxidation stability (>4.9 V), and outstanding ionic conductivity (0.29 mS/cm) at −70 °C.
Enhanced Lithium Metal Anode Performance: Weak F–Li+ coordination in the solvation shell enables Coulombic efficiencies up to 99.7% during lithium plating/stripping at −50 °C, with exchange current densities 10 times higher than O-based systems.
Record Energy Densities: Lithium-metal pouch cells utilizing these electrolytes achieve energy densities exceeding 700 Wh/kg at room temperature and ~400 Wh/kg at −50 °C, even with minimal electrolyte usage (<0.5 g Ah−1).
Implications for Future Energy Storage
These hydrofluorocarbon (HFC) electrolytes circumvent limitations of traditional coordination chemistry, offering a scalable solution for batteries operating in extreme cold—critical for applications in electric vehicles, aerospace, and renewable energy storage. The breakthrough underscores fluorine’s potential to revolutionize solvent design for next-generation high-performance batteries.

Zhao Qing introduced that the key to achieving the dissolution of lithium salts through fluorine coordination lies in regulating the electron density of fluorine atoms and the steric hindrance of solvent molecules. The newly developed lithium battery has advantages such as high specific energy and low-temperature tolerance.
Source: Nature, DOI: 10.1038/s41586-026-10210-6

